 In the harmonic limit, all degrees of freedom are treated harmonically. Parameters if the entropy or free energy are desired, such as the geometryĪnd symmetry number. 
The IdealGasThermo class would generally be called after an energy Returns the Gibbs free energy, in eV, in the ideal gasĪpproximation at a specified temperature (K) and pressure (Pa). get_gibbs_energy ( temperature, pressure, verbose = True ) ¶ Returns the entropy, in eV/K, in the ideal gas approximationĪt a specified temperature (K) and pressure (Pa). get_entropy ( temperature, pressure, verbose = True ) ¶ Returns the enthalpy, in eV, in the ideal gas approximationĪt a specified temperature (K). get_enthalpy ( temperature, verbose = True ) ¶ If False (default), a ValueError will be raised ifĪny imaginary frequencies remain after the 3N-5/3N-6 cut. Will not be included in the calculation of the thermochemical If True, any imaginary frequencies present after the 3N-5/3N-6 cut (0 for molecules in which all electronsĪre paired, 0.5 for a free radical with a single unpaired electron,ġ.0 for a triplet with two unpaired electrons, such as O_2.) ignore_imag_modes bool Cramer “Essentials of Computational Chemistry”, See, for example, Table 10.1 andĪppendix B of C. Used to calculate rotational moments of inertia and molecular mass symmetrynumber integer In ‘atoms’ or if the user desires the entire list of vibrationsĮxtra inputs needed for entropy / free energy calculations: atoms an ASE atoms object (Not needed if an atoms object is supplied The number of atoms, used along with ‘geometry’ to determine how (if potentialenergy is unspecified, then the methods of thisĬlass can be interpreted as the energy corrections) natoms integer The potential energy in eV (e.g., from atoms.get_potential_energy) Geometry of the molecule potentialenergy float geometry ‘monatomic’, ‘linear’, or ‘nonlinear’ If more are specified than are needed, then the lowest Used is automatically calculated by the geometry and the number ofĪtoms. Inputs for enthalpy calculations: vib_energies listĪ list of the vibrational energies of the molecule (e.g., fromĪse._energies). IdealGasThermo ( vib_energies, geometry, potentialenergy = 0.0, atoms = None, symmetrynumber = None, spin = None, natoms = None, ignore_imag_modes = False ) ¶Ĭlass for calculating thermodynamic properties of a moleculeīased on statistical mechanical treatments in the ideal gas IdealGasThermo class supports calculation ofĮnthalpy ( \(H\)), entropy ( \(S\)), and Gibbs free energy ( \(G\)),Īnd has the interface listed below. Translational, rotational, and vibrational degrees of freedom. 
The thermodynamic quantities of ideal gases are calculated by assuming thatĪll spatial degrees of freedom are independent and separable into Likewise, the crystalline solid model depends onĪn accurate phonon density of states this is readily calculated using the The first three cases rely on good vibrational energiesīeing fed to the calculators, which can be calculated with the 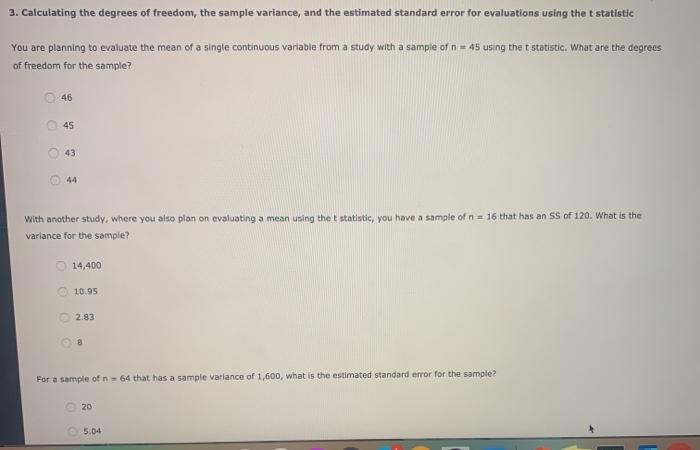
Model (in which a lattice of N atoms is treated as a system of 3N independent Rotational, and the remaining 3N-3 are vibrational), and a crystalline solid Harmonically), the hindered translator / hindered rotor model (used forĪdsorbates, in which two degrees of freedom are translational, one is (generally used for adsorbates, in which all degrees of freedom are treated Rotational degrees of freedom are taken into account), the harmonic limit 
Handled by this module: the ideal-gas limit (in which translational and
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
